You’d think this is one of the easiest science questions ever. And honestly? Most of the time, it is. But if you’ve ever wondered “what is the boiling point of water in Celsius and does it ever change”, things get a little more interesting than just a number.
Let’s start with your instinct.
Quick Gut Check
No calculators, no overthinking:
At what temperature does water boil?
- 50°C
- 100°C
- 150°C
You probably didn’t hesitate. But let’s go a bit deeper than the obvious answer.
The Classic Answer Everyone Knows
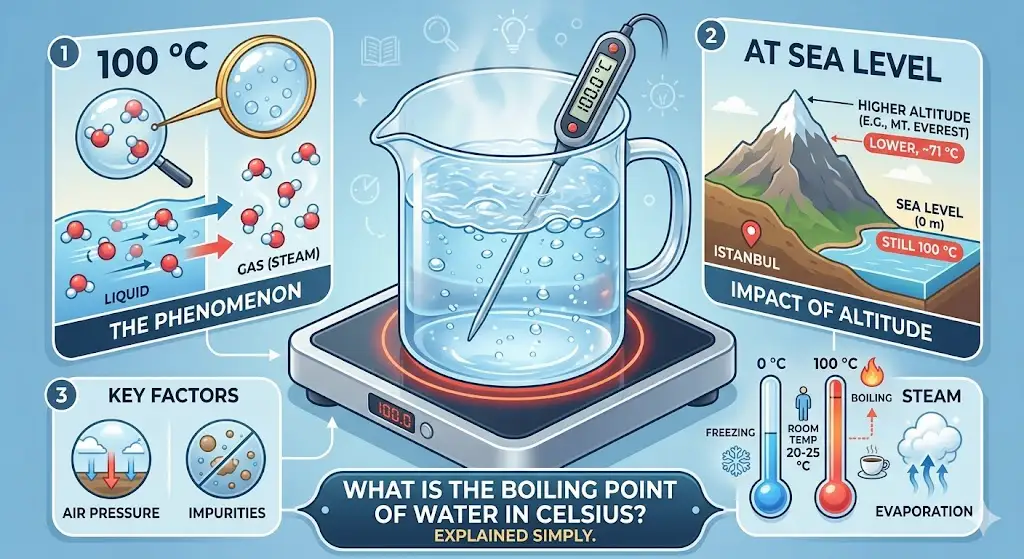
Under normal conditions, water boils at 100°C (degrees Celsius).
That’s the standard answer you’ll see in textbooks, exams, and everyday science facts. It’s based on water reaching its boiling point at standard atmospheric pressure (1 atm) — basically, sea level conditions.
So if someone asks “what temperature does water boil at in Celsius at sea level”, the answer is clean and simple: 100°C.
But Here’s Where It Gets Interesting
Now let me ask you something:
Have you ever boiled water in the mountains?
Because if you have, you might’ve noticed something strange… it boils faster, but your food takes longer to cook.
Why?
Does Water Always Boil at 100°C?
Not exactly. The boiling point changes depending on air pressure.
What Affects the Boiling Point of Water in Celsius
- Higher altitude (lower pressure): water boils below 100°C
- Lower altitude / higher pressure: water boils above 100°C
- Pressure cookers: increase pressure, raising boiling temperature
So if you’re searching “does water always boil at 100 degrees Celsius”, the honest answer is:
Only under standard conditions.
Real-Life Example
Let’s make it practical.
- At sea level → 100°C
- On a mountain → maybe 90–95°C
- Inside a pressure cooker → can go above 100°C
Same water, different behavior. Science doing its thing.
Mini Quiz (Lock It In)
Let’s make sure it sticks:
What is the boiling point of water in Celsius at sea level?
A) 90°C
B) 100°C
C) 120°C
Correct answer: B
Easy — but now you know the hidden twist behind it.
Interesting, Weird, and Everyday Facts
This “simple” topic has some fun layers.
- Water doesn’t just boil — it changes state from liquid to gas
- Bubbles form when vapor pressure equals atmospheric pressure
- On Mount Everest, water can boil at around 70°C
- That’s why cooking takes longer at high altitudes
- The Celsius scale itself was designed using water’s freezing (0°C) and boiling points (100°C)
Here’s a weird one: technically, water can stay liquid above 100°C for a moment under special conditions — it’s called superheating.
Let Me Ask You This
Next time you boil water, think about it:
Are you really just heating water…
or are you watching physics, pressure, and molecular energy all working together in your kitchen?
Why This Still Matters
Even in 2026, people search “what is the boiling point of water in Celsius” because it connects basic science to real life:
- Cooking and food preparation
- Weather and altitude effects
- Scientific experiments
- Engineering and industrial processes
It’s one of those facts that seems basic — until you actually understand it.
And once you do, it’s hard to unsee how often it shows up in everyday life.